|
|
|||||||||||||||
|
Polyphenols and phytochemicals |

|
||||||||||||||
|
What are phytochemicals Classification Polyphenols and cancer Mechanisms of action Enhancing their effect |
|||||||||||||||
What are phytochemicals? Phytochemicals are the gifts from nature, which are responsible for the colour, taste and aroma of plant based foods. As well adding to quality of food, they protect us from environmental and ingested carcinogens and have direct anticancer properties. Strong scientific data has linked their regular intake with a lower risk of premature aging and many other conditions which effect health including:
Classification of phytochemicals There are four major groups of phytochemicals:
1. The Polyphenols Flavonoids:
Phenolic acids
Other non-flavonoid polyphenols
2. Terpenoids 1. Carotenoid terpenoids:
2. Non-carotenoid terpenoids
3. Thiols
Other phytochemicals
Clinical evidence for phytochemicals against cancer Low polyphenol rich diets have been linked to a number of degenerative chronic illnesses such as dementia, high cholesterol, arthritis, skin aging and macular degeneration [Rezai-Zadeh, Maclarty, Elmets]. On the other hand, polyphenol rich diets have been linked to both a reduced risk of developing cancer and improved outcomes after initial treatments. The most notable scientific clinical studies are summaries here: Carotenoids found in leafy green vegetables and
carrots, have been in a number of larger population studies to be linked with a
lower risk of breast cancer as well as ovarian and pancreatic cancers in
smokers. [Hu, Tung, Banim, Chaoyang]. Sulfuraphane and Indol 3 carbinol in broccoli and other cruciferous vegetables, such as cabbage, cauliflower, Brussel sprouts and radishesrich in have been associated with a lowerprostate cancer risk [Joseph]. Broccoli induces the anti-oxidant enzymes glutathione S-transferases which explains why it is particularly beneficial in the 50% of the population carrying a mutated glutathione gene (GSTM1) [Heinen]. Curcuminoids found in in curcurmin, gives turmeric its yellow
colour. Research has also shown that there are low rates of certain types of
cancer in countries where people eat curcumin at levels of about 100 to 200 mg a
day over long periods of time. A clinical trial looked at giving curcumin to
patients with pre cancerous changes in the bowel show that it could stop the
precancerous changes becoming cancer [CRUK]. A number of laboratory studies
shows it can able to kill cancer cells and prevent more from growing. It has
proven effects on breast, prostate, bowel, stomach cancer and skin cancer cells.
A 2007 American study that combined curcumin with chemotherapy to treat
bowel cancer cells in a laboratory showed that the combined treatment killed
more cancer cells than the chemotherapy alone. Tannins
and phenolic acids ingreen tea lower risk of breast, prostate, ovarian and oesophageal
cancer particularly among smokers and alcoholics [Sun, Wu 2006]. High green
teaintake after cancer reduced relapse rates both for breast [Ogunleye] and colorectal
cancer [Zhu]. It also
decreased the abnormal white cell count in 30% of patients with chronic
leukaemia and reduced the levels of several proliferation factors,
as well as PSA among men with
prostate cancer [Shanafelt]. Ellagic acidis
found in pomegranate, which in phase II and randomised studies involving men
with prostate cancer, itslowed the rate PSA progression and other markers of
cancer progression especially if given in a combination of other polyphenol rich
foods as a supplement [Paller, Thomas, Carducci, Pantuck]. Lignans- Women with breast cancer who had the highest serum lignan levels,
reflecting good intake of legumes, cereals, nuts and soya, were reported to have
the lowest risk of death [Buck 2011]. Likewise, a lignan and polyphenol rich
diet was associated with a lower colorectal
cancer relapse rate [Zhu]. Isoflavonesand flavanonesfoundpulses
and soy productsare linked with lower breast and prostate cancer especially if
they had regular intake from childhood [Song-yi]. The largeShanghai Breast
Cancer Survival Study showed that women with the highest intake of the
phytoestrogenic polyphenols isoflavones
and had a 29% lower risk of relapse and death [Boyapati]. Lycopene
found in
colourful fruits and tomatoes has been linked to lower prostate cancer risk [Giovannucci] Flavonoids such as quercetin found in onions, have been shown to reduce theincidence of numerous
cancers particularly those arising from the lung in
smokers [Knekt, Le Marchand]. Anthoxanthins, in dark chocolate, have been reported to lower the risk of colon cancer
[Rodríguez-Ramiro] Phenolic and caffeic acids in coffee has been
shown to reduce the risk of all types of skin cancers [Song, Loftfield] and colon cancer growth in laboratory studies [Kang]. Zeaxanthin rich foods, found leafy green vegetables, reduced the subsequent new skin cancer formation after treatment of a primary lesion in a large Australian study [Heinen].
What are the anti-cancer mechanisms of phytochemicals? 1. Antioxidant properties The most quoted cancer prevention mechanism is via their antioxidant activity, elicited either through direct free radical absorption or through induction of antioxidant enzymes such as superoxide dismutase (SOD), catalase and glutathione via a variety of molecular mechanisms [Reuland, Johnson]. One of these mechanisms is via activation of Nrf2, which switches on genes that code for antioxidant as well as detoxification enzymes [Reuland, Johnson]. Phytochemicals have also been shown to inhibit the conversion of pro-carcinogens to their electrophilic (DNA damaging), particularly the thiol class such as sulforaphane [Gasper, Johnson]. A number clinical studies involving known common carcinogens have highlighted the antioxidant properties of phytochemicals. One experiment involved the known house-hold carcinogen triclocarban, commonly found in detergents and cleaning agents. Healthy cells exposed to triclocarban tend to
mutate into pre-malignant cells, however, the amount and rate of carcinogenesis
was significantly reduced by adding curcumin to the petri disk
culture feeds [Sood]. In another study, volunteers who ate a diet rich in kaempferol were found, on serum and urine analysis, to haveimproved SOD
activity and higher urinary concentration of these polyphenols [Kim]. Rats exposed to cigarette smoke given
indole-3-carbinol, a phytochemical rich in cruciferous vegetables, had a lower
lung cancer rate than those not given idole-3-carbinol [Morse 1990]. Subjects
eating a meal of onions, which increased their serum levels of quercetin,
demonstrated decreased levels of oxidative metabolites including
8-hydroxydeoxyguanosine (8-OHdG) a marker of DNA damage and repair [Boyle, Wu]. A
clinical study from Singapore gave Chinese smokers 170g of watercress a day,
rich in the indole-3-carbinol, and found a similar effect on urinary markers of
DNA damage [Hecht]. Marinating meat in
rosemary and thyme, has been reported to reduced the serum levels of
carcinogenic heterocyclic amines (HCA) by 87% compared to subjects who eat the
meat unseasoned [Smith]. 2. Reducing chronic inflammation. Another key anti-cancer mechanism of phytochemicals appears to be their ability to reduce inappropriate inflammation. It is now well established that inflammation is intimately involved in the cancer process, particularly in the promotion and progression stages of cancer. Inflammation is closely associated with oxidative stress and activation of NF-kappa B family of transcription factors. These factors regulate more than 150 genes involved in mechanisms of cell survival and these target genes are not just pro-inflammatory but also oncogenic. Numerous phytochemicals have been shown to inhibit NF-kappa B signaling particularly the green tea polyphenol epigallocatechin-3-gallate (EGCG), curcumin and bilberries [Karlsen]. 3. Direct biological effects. More recently, it has been discovered that phytochemicals have an affect on several cancer process through modulation of cellular and signalling events involved in growth, invasion and metastasis [Johnson]. Pomegranate, for example, rich in the polyphenol ellagic acid, has been shown to directly inhibit cell growth and induce apoptosis in androgen sensitive and aggressive human prostate cancer cells [Retitig, Malik]. It has also been reported to inhibit processes involved in cancer metastasis in a study involving oestrogen sensitive and resistant breast cancer cell lines showing increased markers of cell adhesion and migration in cancer but not normal cells [Lansky]. In another studying it inhibited a chemokine that attracts breast cancer cells to the bone [Rocha]. Curcumin slows cancer cell growth by blocking the cell cycle, increasing the rate of apoptosis and preventing the invasion and migration of cells [Somasundaram, Zhang, Dorai, Iqbal]. It has also been found to halt the growth of stem cells that give rise to breast cancer without harming normal breast stem cells [Handler]. Curcumin is able to modulate miRNA expression in
breast cancer cells leading to a reduced expression of Bcl-2
[Yang, 2010] and stabilisation of tumour suppressor gene in colorectal
cancercell lines[Mudduluru].
Green tea, rich in
epigallocatechingallate (EGCG), has demonstrated significant reduction of
several factors that promote cancer cell growth, de-differentiation and
angiogenesis [Shanafelt, Pietinen, Voorrips]. It has also been shown to block ornithine
decarboxylase, an enzyme which signals cells to proliferate faster and bypass
apoptosis [Yang, Liao). Broccoli sparks numerous genetic changes, activating cancer suppressor
genes and switching off promotion genes [Moysich]. Resveratrol has demonstrated
epigenetic regulatory properties which influence regulate
cell survival and apoptosis in prostate cancer by
global modulation of gene expression through deacetylation of FOXO transcription
factor [Chen]. Can concentrating foods into supplements enhance their benefits? If
certain foods have anti-cancer effects, then it is not unreasonable to
hypothesise that concentrating them into a pill may be a good way to supplement
individuals with a poor diets or further enhance the benefits in those who’s
diets are already adequate. There are two main categories supplements
commercially available. The first involves chemicals extracted from food or made
synthetically such as minerals and vitamins, the second involves purifying and
concentrating whole foods:
The negative effects of vitamin E and beta carotene were once again demonstrated in the ATBC study which found them to increase lung cancer risk, although subsequent analysis showed men with pre-intervention low plasma levels of beta-carotene had a lower prostate cancer risk following supplementation and those with high levels had a higher risk, particularly in smokers[Heinonen]. This u-shaped distribution of risk was also observed in the EPIC study where those with folate-deficient diets and those with the highest intake both had a higher risk of cancer [Chuang]. These data have prompted organisation such as the National Cancer Institute to issue statements stating that long term vitamin and mineral supplements should ideally be given to correct a known deficiency [Greenwald] which is rarely routinely detected unless individuals have self funded micro-nutrient analysis (link). Phytochemical rich whole food supplements:
A recent study reported that a supplement containing several phytochemical rich foods including resveratrol, citrus bioflavonoids and chamomile called Phyto-v had significant anti-viral properties and significantly improved recovery from long covid....read more Sleep Emerging from the covid data a phytochemical rich, non-sedating sleeping aid has been developed which has been shown to improve sleep, improve day time alertness without a hangover effect - this is called phytonight+ Other symptoms A number of other RCT’s involving whole food phytochemical rich supplement have demonstrated benefits for some of the distressing symptoms common after cancer treatments such as fatigue [Barton] and urinary infections [Bonetta]. As part of the COVID study mentioned above, patients reported their troublesome peripheral neuropathy improved or resolved after taking Phyto-v. A new double blind RCT is planned but in the mean time people are reporting anecdotal benefit from this supplement. Prostate Cancer
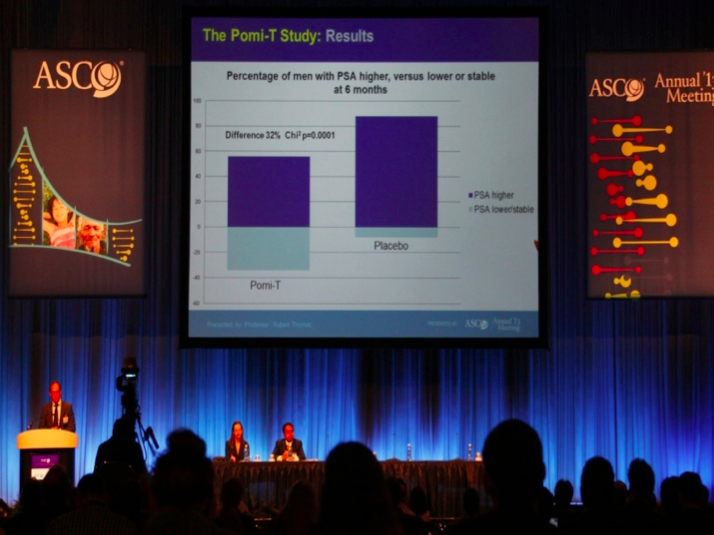
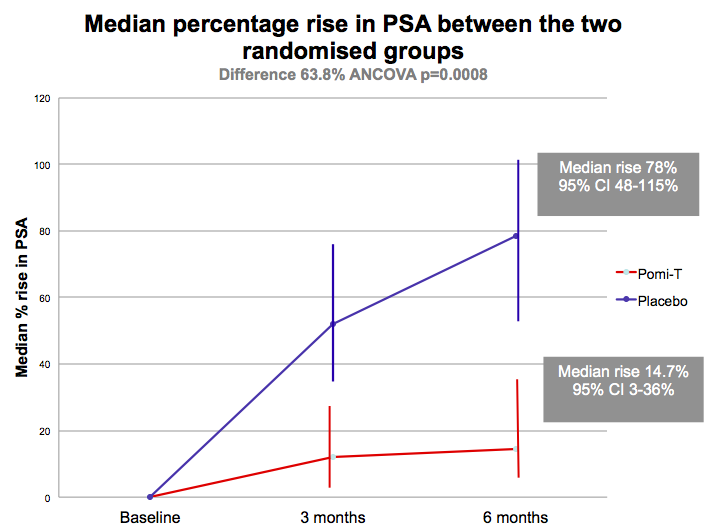
It was well tolerated, apart from some mild loosening
of the bowels in 10% of men, and there was no effect on testosterone levels. At 6 months, significantly more men
opted to remain on surveillance rather than proceeding to expensive
radiotherapy, surgery or medical castration which can cause unpleasant effects
such as depression, hot flushes, weight gain, osteoporosis, erectile dysfunction
[Thomas]. The latest UK nation trial involving men with prostate cancer In 2024 a new phytochemical rich supplement has been developed by the same research team for the latest and largest nutritional intervention study in the World It has enhanced the phytochemical content via additional extracts and two more ingredients which had proven benefits for prostate symptoms. It is known as Yourphyto and the it is being investigated along side a probiotic/vitamin D complex in a major UK double blind randomised trial starting September 2023. and finished recruiting in August 2024....read more Conclusion There is increasingly convincing evidence to show
that phytochemicals not only do they improve our daily lives by helping our food
taste, smell and look appetizing they reduce our risk of cancer but help people
living with and beyond treatments. Living well programmes, slowly being
introduced in the UK, are beginning to highlight the importance of phytochemical
rich diets, along side other lifestyle factors largely being driving by the
National Survivorship Initiative and guidelines from influential organisations
such as American Society of Clinical Oncology [ASCO]. Going a step further and
concentrating these foods or extracted elements of these foods into nutritional
supplements gives an opportunity to boost their beneficial anti-cancer effects
but have their pitfalls. Studies of concentrated minerals, vitamins and
phytoestrogenic supplements have reported detrimental effects although no study
has reported detrimental effects of whole non-phytoestrogenic food supplements
and some significant advantages. Despite these potential benefits and reports
that over 60% of patients living with and beyond cancer take nutritional
supplements, oncologist have been reluctant to discussing their pros and cons due to a lack of
RCT from academic institutions [Uzzo,
Bauer]. Hopefully this trend will
change particularly following the
success of the ongoing YourPhyto
study.
References 1. World Cancer Research Fund/American Institute for Cancer Research. Food, Nutrition and the Prevention of Cancer: A Global Perspective. AIRC: Washington; 2007. 2. Key TJ. Fruit and vegetables and cancer risk. British Journal of Cancer 2011;104: 6Ð11. 3. Block G, Patterson B and Subar A. Fruit, vegetables and cancer prevention: a review of the epidemiological evidence. Nutrition and Cancer 1992;18(1): 1Ð29. 4. Scalbert A, Johnson I and Satlmarsh M. Polyphenols: antioxidants and beyond. American Journal of Clinical Nutrition 2005;81(1): 215S-217S. 5. Bauer CM et al. Prevalence and correlates of vitamin and supplement usage among men with prostate cancer. Integrative Cancer Ther 2012;11(2): 83-9. 6. Uzzo RG et al. Prevalence and patterns of self-initiated nutritional supplementation in men at high risk of prostate cancer. British J of Urology Int 2004;93(7): 955-60. 7. Hu F et al. Carotenoids and breast Cancer risk: a meta-analysis and meta-regression. Breast Cancer Research and Treatment 2012;131(1): 239-53. 8. Tung K et al. Association of dietary vitamin A, carotenoids and other antioxidants with the risk of ovarian cancer. Cancer Epi, Biomarkers & Preven 2005;14: 669. 9. Banim PJ et al. Dietary antioxidants and the aetiology of pancreatic cancer: a cohort study using data from food diaries and biomarkers. Gut 2012;62(10): 1489-96. 10. Chaoyang L et al. Serum alpha-carotene concentrations and the risk of death amongst US adults. Archives of Internal Medicine 2011;171(6): 507-515. 11. Joseph MA et al. Cruciferous vegetables, genetic polymorphisms and prostate cancer risk. Nutrition and Cancer 2004;50(2): 206-213. 12. Song-Yi , et al. Legume and isoflavone intake and prostate cancer risk: The Multi-ethnic Cohort Study. International Journal of Cancer. 2008;123(4): 927-932. 13. Giovannucci E, Rimm EB, Liu Y, et al. A prospective study of tomato products, lycopene and prostate cancer risk. J of the Nat Can Inst 2002;94: 391-98. 14. Knekt P et al. (1997) Dietary flavonoids and the risk of lung cancer and other malignant neoplasms. American Journal of Epidemiology 1997;146: 223Ð230. 15. Le Marchand L, Murphy SP, Hankin JH, et al. Intake of flavonoids and lung cancer. Journal of the National Cancer Institute 2000;92: 154 Ð160. 16. Rodr’guez-Ramiro D, et al. Cocoa-rich diet prevents azoxymethane-induced colonic preneoplastic lesions in rats by restraining oxidative stress and cell proliferation and inducing apoptosis. Molecular Nutrition & Food Research 2011;55: 1895-99. 17. Sun CL, Yuan JM, Koh WP, et al. Green tea and cancer risk: The Singapore Chinese Health Study. Carcinogenesis 2007;28(10): 2143-2148. 18. Wu LL et al. Urinary 8-OHdG: a marker of oxidative stress to DNA and a risk factor for cancer, atherosclerosis and diabetics. Clinica Chimica Acta 2004;339(1-20): 1-9. 19. Song F, Qureshi A and Han J. Increased caffeine intake is associated with reduced risk of basal cell carcinoma of the skin. Cancer Research 2012;72(13): 3282-89. 20. Loftfield E et al. Coffee drinking and cutaneous melanoma risk in the NIH-AARP diet and health study. Journal of the National Cancer Institute 2015;107(2): 1-9. 21. Pierce JP, et al. Influence of a diet very high in vegetables, fruit, and fiber and low in fat on prognosis following treatment for breast cancer: the Women's Healthy Eating and Living (WHEL) randomized trial. Journal of the American Medical Association 2007;298(3): 289-298. 22. Buck K, Vrieling A, Zaineddin AK, et al. Serum enterolactone and prognosis of post-menopausal breast cancer. Journal of Clinical Oncology 2011;29(28): 3730-38. 23. Zhu Y, Wu H, Wang PP, et al. Dietary patterns and colorectal cancer recurrence and survival: a cohort study. British Medical Journal Open 2013;3(2): e002270. 24. Boyapati SM. Soy food intake and breast cancer survival: a follow up of the Shanghai Breast Cancer Study. Breast Cancer Research and Treatment 2005;92: 117. 25. Ogunleye AA, Xue F and Michels KB. Green tea and breast cancer risk of recurrence: A meta-analysis. Breast Cancer Research and Treatment 2010;119(2): 477. 26. Shanafelt TD, et al. Phase I trial of daily oral polyphenon E (green tea extract) in patients with asymptomatic stage 0-II chronic lymphatic leukaemia. JCO 2009;27(23): 3808Ð3814. 27. Ornish D, Weidner G, Fair WR, et al.; Intensive lifestyle changes may affect the progression of prostate cancer. Journal of Urology 2005;174: 1065-1070. 28. Pantuck AJ et al. Phase II study of pomegranate juice for men with rising PSA following surgery or radiation for prostate cancer. Journal of Urology 2005;173: 225Ð226. 29. Heinen MM, Hughes MC, Ibiebele TI, et al. Intake of antioxidant nutrients and the risk of skin cancer. European Journal of Cancer 2007;43(18): 2707-2716. 30. Swann R et al. The DietCompLf study: A prospective cohort study of breast cancer survival and phytoestrogen consumption. Maturitas 2013;75: 232-240. 31. Reuland DJ, et al. Upregulation of phase II enzymes through phytochemical activation of Nrf2 protects cardiomyocytes against oxidant stress. Free Radical Biol and Med 2013;56: 102Ð111. 32. Johnson I. Phytochemicals and cancer. Proceedings of the Nutrition Society 2007;66: 207-215. 33. Gasper AV et al. Glutathione S-transferase M1 polymorphism and metabolism of sulforaphane from broccoli. Am Journal of Clin Nut 2005;82: 128-91. 34. Sood et al. Induction of Human Breast Cell Carcinogenesis by Triclocarban and Intervention by Curcumin. Biochem and Biophys Res Coms 2013;438(4): 600-6. 35. Kim HY et al. Effects of phenol-depleted and phenol-rich diets on blood markers of oxidative stress, and urinary excretion of quercetin and kaempferol in healthy volunteers. Journal of the American College of Nutrition 2003;22(3): 217-223. 36. Morse MA, LaGreca SD, Amin SG, et al. Effects of indole-3-carbinol on lung tumorgenesis and DNA methylation in mice. Cancer Research 1990;50: 2613-2627. 37. Boyle SP et al. Absorption and DNA protective effects of flavonoid glycosides from an onion meal. European Journal of Nutrition 2000;39: 213Ð223. 38. Sandhir R. Quercetin improvesc mitochondrial dysfunctions induced by 3-nitropropionic acid: Implications in Huntington's disease. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2013; 1832 (3): 421-430. 39. Hecht SS, et al. Effects of cruciferous vegetable consumption on urinary metabolites of the tobacco-specific lung carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone in Singapore Chinese. Cancer Epidemiology, Biomarkers & Prevention 2004; 13(6): 997-1004 .40. Smith JS and The Food Safety Consortium. Brush on the marinade, hold off the cancerous compounds. ScienceDaily 2007;June 28. 41. Carlsen MH, et al. The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide. Nutrition Journal 2010;9:3 1475-2891-9-3. 42. Malik A et al. Pomegranate fruit juice for chemoprevention and chemotherapy of prostate cancer. Proceedings of the National Academy of Sciences 2005;102: 14813Ð14818. 43. Lansky EP et al. Possible synergistic prostate cancer suppression by anatomically discrete pomegranate fractions. Investigational New Drugs 2005;23: 11Ð20. 44. Rocha A et al. Pomegranate juice s inhibit cell and molecular processes critical for metastasis of breast cancer. Breast Cancer Research and Treatment 2012;136(3): 647-58. 45. Somasundaram S, Edmund NA, Moore DT et al. Curcumin inhibits chemotherapy-induced apoptosis in models of cancer. Cancer Research 2002;62(13): 3868-3875. 46. Park EJ et al. The pharmacology of resveratrol in animals and humans. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2015; 1852 (6); 1071-1113 47. Butterfield DA and Keller J. Antioxidants and antioxidant treatment in disease. Biochimica et Biophysica Acta 2012; 1822: 615 48. Zhang HN, Yu CX, Chen WW, et al. Curcumin down regulates gene NKX3.1 in prostate cancer cell lines (LNcaP). Acta Pharmacologica Sinica 2007;28(3): 423-430.49. Dorai T. Therapeutic potential of curcumin in human prostate cancer. - inhibits tyrosine kinase activity of the epidermal growth factor receptor. Molecular Urol 2000;4(1): 1-6. 50. Iqbal M, et al. Dietary supplementation of curcumin enhances antioxidant phase II metabolosing enzymes in mice. Pharmacology & Toxicology 2003;92(1); 33-38. 51. Handler N et al. Synthesis of noval curcumin analogues and their evaluation as selective cyclooxygenase-1 inhibitors. Chemical & Pharmaceutical Bulletin 2007;55(1): 64-71. 52. Yang CS, Maliakal P and Meng X. Inhibition of carcinogenesis by tea. Annual Review of Pharmacology and Toxicology 2002;42: 25-54. 53. Mudduluru G et al. Curcumin regulates miR-21 expression and inhibits invasion and metastasis in colorectal cancer. Bioscience Reports 2011;31(3): 185-97. 54. Pietinen P, et al. Diet and risk of colorectal cancer in a cohort of Finnish men. Cancer Causes and Control 1999;10(5): 387-96. 55. Voorrips LE et al. Veg and fruit consumption and risks of colon in a prospective cohort study: The Netherlands Cohort Study on Diet and Cancer. Am J of Epi 2000 1;152(11): 1081-92. 56. Liao J, (2004). Inhibition of lung carcinogenesis and effects on angiogenesis and apoptosis in mice given green tea. Nutrition and Cancer 2004;48(1): 44-53. 57. Harris HR, Orsini N and Wolk A. Vitamin C and survival among women with breast cancer: a metanalysis. European Journal of Cancer 2014;50(7): 1223-1231. 58. Hercberg S , et al. The SU.VI.MAX Study: a RCT of the health effects of antioxidant vitamins and minerals. Archives of Internal Medicine 2004;164(21): 2335-42. 59. Meyer F et al. Antioxidant vit & mineral supplementation & prostate cancer prevention in the SU.VI.MAX trial. Int J of Can 2005;116(2): 182-6. |
|||||||||||||||
 The majority of studies, to date, have evaluated
extracted chemicals such as
The majority of studies, to date, have evaluated
extracted chemicals such as