Avastin is a biological agent with a generic name called bevacizumab. This information sheet provides a brief
introduction to Avastin and explains the common side effects you may experience.
This does not mean you will definitely get them. It is also possible you may
experience a side effect not mentioned here.
Avastin is usually used in conjunction with a chemotherapy
agent called 5 Fluorouracil/folonic acid or Taxol
sometimes given with irinotecan
capecitabine or vinorelbine. It is generally given to patients with bresat or
colorectal cancer but there is emerging evidence of benefit
with other cancers.
The exact combination would have been explained to you by your doctor and you
should also refer to the specific information and advice sheet relating to these
other drugs.
WHAT IS AVASTIN?
Avastin is currently licensed as a treatment for metastatic colorectal cancer.
This means that cancer cells have spread from the original site in the colon
or rectum 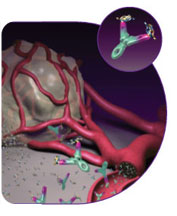 to
another part of the body (most often the liver). Avastin
is not a chemotherapy or a hormone but a class of agents known either as immunotherapy or
biological therapies. More specifically they are a group of large proteins
called monoclonal
antibody (Mab). These agents are similar to the antibodies the
body's own immune system normally makes when we have a bacterial or viral infection but in this case it has been made in a
laboratory and attacks specific targets on cancer cells.
In the case of Avastin the target (otherwise called the antigen) is a protein called
Vascular Epidermal Growth Factor (VEGF). Avastin binds to
VEGF, rendering
it unable to then bind with its receptor which in turn by blocking the formation
of new blood vessels then interferes with the growth of the tumour (see below). to
another part of the body (most often the liver). Avastin
is not a chemotherapy or a hormone but a class of agents known either as immunotherapy or
biological therapies. More specifically they are a group of large proteins
called monoclonal
antibody (Mab). These agents are similar to the antibodies the
body's own immune system normally makes when we have a bacterial or viral infection but in this case it has been made in a
laboratory and attacks specific targets on cancer cells.
In the case of Avastin the target (otherwise called the antigen) is a protein called
Vascular Epidermal Growth Factor (VEGF). Avastin binds to
VEGF, rendering
it unable to then bind with its receptor which in turn by blocking the formation
of new blood vessels then interferes with the growth of the tumour (see below).
HOW DOES AVASTIN
WORK?
When
cancer cells spread to another part of the body they try to form a lump or
tumour 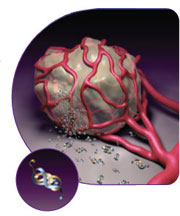 mass. In order to do this, they need
to rapidly stimulate the local blood vessels and capillaries to grow into the
tumour mass in order to nourish the cancer cells with food and oxygen (a process
called angiogenesis). They achieve
this by releasing a chemical into the surrounding tissues called Vascular
Epidermal Growth Factor (see adjacent picture). Avastin, being a monoclonal
antibody which attaches to circulating VEGF, effectively blocks its
ability to bind to its receptors in the tissues of the body, most importantly
those surrounding tumours. Avastin, therefore interferes with the tumours
ability to recruit new blood vessels reducing their ability to grow and spread to other areas of the
body. As this process is universal to most bowel tumours, there is no
requirement to perform extra laboratory tests on the cancer cells prior to Avastin
therapy. There is also evidence that Avastin enhances the effect of
chemotherapy. It is thought they make tumour vessels less "leaky" and
so allows chemotherapy to reach the tumour more effectively. mass. In order to do this, they need
to rapidly stimulate the local blood vessels and capillaries to grow into the
tumour mass in order to nourish the cancer cells with food and oxygen (a process
called angiogenesis). They achieve
this by releasing a chemical into the surrounding tissues called Vascular
Epidermal Growth Factor (see adjacent picture). Avastin, being a monoclonal
antibody which attaches to circulating VEGF, effectively blocks its
ability to bind to its receptors in the tissues of the body, most importantly
those surrounding tumours. Avastin, therefore interferes with the tumours
ability to recruit new blood vessels reducing their ability to grow and spread to other areas of the
body. As this process is universal to most bowel tumours, there is no
requirement to perform extra laboratory tests on the cancer cells prior to Avastin
therapy. There is also evidence that Avastin enhances the effect of
chemotherapy. It is thought they make tumour vessels less "leaky" and
so allows chemotherapy to reach the tumour more effectively.
HOW IS AVASTIN
GIVEN?
Avastin cannot be taken orally as it would be destroyed by your stomach. It
is therefore infused into a vein usually in your arm or hand. The first treatment with Avastin will normally take 90 minutes,
and will be given after your chemotherapy. After this you’ll be given one ‘maintenance’ dose
usually every 2 weeks. Over time, the infusion time may decrease
to around 60 and then 30 minutes once your doctor is happy that you
have no problems. Occasionally, as with all protein based drugs it is possible to
get
an allergic reaction (<3% of cases). The nurses will therefore be checking how you are
feeling and measuring your breathing, pulse and blood pressure regularly. Sometimes, in
response to mild reaction, the infusion may have to be slowed down over several hours.
Rarely if the allergic reaction is prominent it has to be stopped altogether. To avoid a mild reaction
paracetamol and an antihistamine
are sometimes given before the infusion.
As mentioned Avastin is usually given in
conjunction will chemotherapy the administration schedule and side effect also
depend on these drugs.
As with other therapies,
your doctors will also check your blood before treatment, to ensure adequate
function of your bone marrow, kidneys and liver.
normally take 90 minutes,
and will be given after your chemotherapy. After this you’ll be given one ‘maintenance’ dose
usually every 2 weeks. Over time, the infusion time may decrease
to around 60 and then 30 minutes once your doctor is happy that you
have no problems. Occasionally, as with all protein based drugs it is possible to
get
an allergic reaction (<3% of cases). The nurses will therefore be checking how you are
feeling and measuring your breathing, pulse and blood pressure regularly. Sometimes, in
response to mild reaction, the infusion may have to be slowed down over several hours.
Rarely if the allergic reaction is prominent it has to be stopped altogether. To avoid a mild reaction
paracetamol and an antihistamine
are sometimes given before the infusion.
As mentioned Avastin is usually given in
conjunction will chemotherapy the administration schedule and side effect also
depend on these drugs.
As with other therapies,
your doctors will also check your blood before treatment, to ensure adequate
function of your bone marrow, kidneys and liver.
ARE THERE ANY SIDE EFFECTS WITH AVASTIN?
When
given with chemotherapy the side effect normally relate to the chemotherapy -
you should also have been given an information sheet relating to your
chemotherapy. Avastin in
general, is well tolerated, and does not make these worse. Avastin may, however, have some mild side effects on its own which may occur in addition to those
caused by chemotherapy:-
The early effects are rare (<3%) and related to an
"allergic" reaction. If they do occur, it is often while the drug is
being infused or immediately after. Potential side effects include:
ARE THERE ANY RISKS WITH AVASTIN?
Some patients experience an increase in their blood pressure and may need to take tablets to lower it.
Having high blood pressure does not mean you are not eligible for Avastin
provided it is controlled before treatment. All patients on Avastin will have
their blood
pressure monitored during therapy.
The amount of protein in your urine may rise higher than normal
(particularly if your blood pressure is also raised) – your medical team will check your urine from time to time.
If the protein continues at a high level then Avastin may have to be stopped.
There is a small risk of gastrointestinal perforation or
bleeding from the bowel (<2%). Before treatment with Avastin, your doctor
will therefore ask you whether you have a history of stomach complaints such as active gastric ulcers or colitis.
Avastin may adversely affect wound healing. Therapy should
therefore, not be initiated for at least 28 days following major surgery.
Likewise, if an elective operation is necessary, Avastin should be withdrawn until
healing is complete.
Minor nose bleeds lasting less than 5 minutes requiring no
medical intervention is known to occur in 20-30% of patients.
In patients with bowel cancer
<3% had a bleed from their tumours. If you notice blood in your stool
or vomit or have abdominal pains please report this to your medical team.
There is often an increased risk of blood clots in patients with
cancer particularly whilst on chemotherapy, this risk increases slightly further
with Avastin therapy. It is important therefore to keep as active and mobile as feasible
and to report any chest pain, unexplained breathlessness or dizziness to your
medical team.
WHY HAVE YOU
BEEN RECOMMENDED AVASTIN?
Unfortunately, by the time
you are to be considered for Avastin your disease has spread from the primary
site in the bowel and is progressing. The aim
of treatment now is not to cure, but to control specific
symptoms caused by the
tumour. This is sometimes known as palliative
treatment. It is hoped that treatment improves your quality of life;
therefore the side effects from the treatment should not outweigh the benefits
of shrinking the tumour.
HOW WILL YOU
KNOW TREATMENT IS WORKING?
Your doctor would
require evidence that treatment is helping particularly after a 2-3 months. This
can range from a number of sources including; an improvement in a specific
symptom such as pain, a shrinkage of a lump on examination, improvements in a
blood test such as the CEA (cancer embryonic antigen) or often evidence from
repeat X-rays and scans such as CT or MRI. If there is no palliative benefit by
this time treatment should be stopped as it may be doing more harm than good.
If there is evidence of response,
treatment will continue provided continuing palliative benefit is achieved. The
Avastin may well continue after the chemotherapy has finished but the precise
duration of treatment depends on a close dialogue between yourself and your
oncologist.
HOW LONG WILL YOU TAKE AVASTIN?
As mentioned above if there is no palliative or objective benefit, treatment
should be stopped as soon as this is known - usually after 2-3 cycles of
chemotherapy. If there is a benefit however treatment may continue for as long
as there is a response.
OTHER LIFESTYLE QUESTIONS RELATING TO
AVASTIN
-
Can I drink alcohol while taking Avastin? -
Yes, but in moderation.
-
Can I work while taking Avastin? -
You may continue to work, providing you feel fit enough to do
so.
-
Can I drive while taking Avastin? -
Yes, there is no evidence that Avastin affects driving ability.
-
Can I exercise
while taking Avastin - Its a misconception that exercise should be avoided
following a diagnosis of cancer or during treatment. Light exercise can help
fight fatigue, reduce the risk of blood clots and improve mood.
-
Will I have to change what I eat or drink? -
No, you can eat and drink as normal.
-
The chemotherapy
that you are taking may cause hair loss but Avastin will not make
this worse.
-
Avastin does not affect your sex drive and you should be
able to continue sexual relations. If you are a fertile male or female
contraceptive precautions should be taken.
-
You must
not become pregnant whilst on Avastin or for 6 months after your
last dose, as it may cause irreversible harm to your unborn baby. If you suspect
you are pregnant inform your doctors immediately.
-
Don’t breastfeed while taking Avastin or for 6 months after treatment is stopped.
-
If you need to visit your GP for any other reason tell him or her you are taking Avastin
-
You should not stop taking other prescribed medication without discussing
it with your doctor. Likewise discuss first before taking purchased over the counter
drugs, including herbal remedies.
|

 to
another part of the body (most often the liver).
to
another part of the body (most often the liver).  mass. In order to do this, they need
to rapidly stimulate the local blood vessels and capillaries to grow into the
tumour mass in order to nourish the cancer cells with food and oxygen (a process
called angiogenesis). They achieve
this by releasing a chemical into the surrounding tissues called Vascular
Epidermal Growth Factor (see adjacent picture). Avastin, being a monoclonal
antibody which attaches to circulating VEGF, effectively blocks its
ability to bind to its receptors in the tissues of the body, most importantly
those surrounding tumours. Avastin, therefore interferes with the tumours
ability to recruit new blood vessels reducing their ability to grow and spread to other areas of the
body. As this process is universal to most bowel tumours, there is no
requirement to perform extra laboratory tests on the cancer cells prior to Avastin
therapy. There is also evidence that Avastin enhances the effect of
chemotherapy. It is thought they make tumour vessels less "leaky" and
so allows chemotherapy to reach the tumour more effectively.
mass. In order to do this, they need
to rapidly stimulate the local blood vessels and capillaries to grow into the
tumour mass in order to nourish the cancer cells with food and oxygen (a process
called angiogenesis). They achieve
this by releasing a chemical into the surrounding tissues called Vascular
Epidermal Growth Factor (see adjacent picture). Avastin, being a monoclonal
antibody which attaches to circulating VEGF, effectively blocks its
ability to bind to its receptors in the tissues of the body, most importantly
those surrounding tumours. Avastin, therefore interferes with the tumours
ability to recruit new blood vessels reducing their ability to grow and spread to other areas of the
body. As this process is universal to most bowel tumours, there is no
requirement to perform extra laboratory tests on the cancer cells prior to Avastin
therapy. There is also evidence that Avastin enhances the effect of
chemotherapy. It is thought they make tumour vessels less "leaky" and
so allows chemotherapy to reach the tumour more effectively.
 normally take 90 minutes,
and will be given after your chemotherapy. After this you’ll be given one ‘maintenance’ dose
usually every 2 weeks. Over time, the infusion time may decrease
to around 60 and then 30 minutes once your doctor is happy that you
have no problems. Occasionally, as with all protein based drugs it is possible to
get
an allergic reaction (<3% of cases). The nurses will therefore be checking how you are
feeling and measuring your breathing, pulse and blood pressure regularly. Sometimes, in
response to mild reaction, the infusion may have to be slowed down over several hours.
Rarely if the allergic reaction is prominent it has to be stopped altogether. To avoid a mild reaction
paracetamol and an antihistamine
are sometimes given before the infusion.
As mentioned Avastin is usually given in
conjunction will chemotherapy the administration schedule and side effect also
depend on these drugs.
As with other therapies,
your doctors will also check your blood before treatment, to ensure adequate
function of your bone marrow, kidneys and liver.
normally take 90 minutes,
and will be given after your chemotherapy. After this you’ll be given one ‘maintenance’ dose
usually every 2 weeks. Over time, the infusion time may decrease
to around 60 and then 30 minutes once your doctor is happy that you
have no problems. Occasionally, as with all protein based drugs it is possible to
get
an allergic reaction (<3% of cases). The nurses will therefore be checking how you are
feeling and measuring your breathing, pulse and blood pressure regularly. Sometimes, in
response to mild reaction, the infusion may have to be slowed down over several hours.
Rarely if the allergic reaction is prominent it has to be stopped altogether. To avoid a mild reaction
paracetamol and an antihistamine
are sometimes given before the infusion.
As mentioned Avastin is usually given in
conjunction will chemotherapy the administration schedule and side effect also
depend on these drugs.
As with other therapies,
your doctors will also check your blood before treatment, to ensure adequate
function of your bone marrow, kidneys and liver.